Medical devices are crucial for diagnosing, monitoring, and improving patient health, but every device has a finite lifespan. Whether due to expiration, contamination, or technological obsolescence, these tools eventually need to be removed from service. The responsible destruction of medical equipment not only protects patients and healthcare workers but also ensures regulatory compliance and helps safeguard the environment.
The pathway a device takes post-destruction is highly regulated and driven by the need to prevent improper reuse, data leaks, and environmental harm. Understanding what happens to medical devices after they are destroyed reveals a complex process that balances safety, efficiency, and sustainability.
For healthcare providers and organizations, compliance with disposal regulations is essential. It can be the difference between maintaining a clean bill of legal health and facing fines, license suspension, or reputational damage. Moreover, as the healthcare sector moves toward greater eco-consciousness, the disposal and destruction of medical devices are increasingly affecting hospital sustainability strategies.
Unregulated, improper disposal can result in significant waste problems and even legal consequences. For a deeper look at the legal risks of skipping vital steps in device destruction, industry watchdogs and government websites offer practical insights and case studies.
Why Medical Devices Are Destroyed
Devices are destroyed for multiple reasons. They may be past their expiration date, which poses risks to patients if the device’s materials degrade or its functionality declines. Contamination with biohazards or infectious materials requires immediate, irreversible destruction to eliminate the risk of disease transmission. Additionally, rapid innovation in medical technology often renders older devices obsolete; outdated devices may not meet regulatory standards or support the latest safety protocols. When these scenarios arise, removal and destruction become non-negotiable steps. According to the Centers for Disease Control and Prevention (CDC), strict adherence to disposal protocols is fundamental to public health.
Methods of Medical Device Destruction
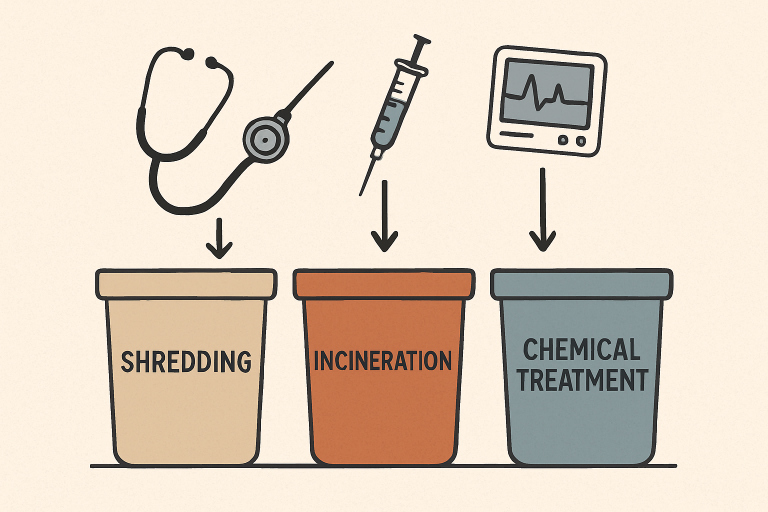
The method used to destroy medical devices depends on the device type, its contents, and regulatory mandates. The most common strategies include the following:
- Shredding: Large medical devices and electronics are physically shredded into small, unrecognizable fragments, preventing reuse or reconstruction.
- Incineration: High heat is used to reduce devices, especially those contaminated with biological materials, to ashes, completely destroying any hazardous material.
- Chemical Treatment: Some items require chemical neutralization to render toxins or biohazards harmless before the remaining waste can be discarded safely.
Healthcare facilities must choose the appropriate destruction method by consulting industry guidelines and adhering to regional and national regulations.
Environmental Considerations
Improperly disposed medical device waste can expose communities to pollutants and chemicals. To address this, hospitals and clinics increasingly invest in recycling and reprocessing programs. Reprocessing, which includes sterilizing and refurbishing devices labeled “single-use,” has become especially popular. This process extends device life, reduces landfill waste, and often leads to significant cost savings for healthcare systems. For example, reports from 2021 show that U.S. facilities reprocessed enough medical devices to divert over 20 million pounds of waste from landfills.
Data Security in Device Disposal
Modern medical devices often contain digital storage or embedded chips that hold sensitive patient data. Ensuring patient confidentiality and protecting data security during disposal are top priorities. Healthcare organizations undertake several measures, including:
- Data Wiping: Removal of all stored information through software-based solutions or memory destruction.
- Physical Destruction: Devices are rendered inoperable through shredding or crushing to prevent data retrieval.
- Documentation: Transparent recordkeeping proves compliance with legal and regulatory requirements for device and data destruction.
Failure to manage data exposure risks during device destruction can lead to breaches that violate the Health Insurance Portability and Accountability Act (HIPAA) and carry significant financial and reputational consequences for organizations.
Regulatory Compliance
Medical device disposal is subject to rigorous regulations managed by entities such as the U.S. Food and Drug Administration (FDA) and the Occupational Safety and Health Administration (OSHA). These govern not only how devices are destroyed but also how waste is tracked and reported. Regulations help healthcare entities minimize environmental and public health risks and provide standard procedures for handling contaminated or hazardous waste. Access the FDA’s full guidance for disposing of contaminated devices on their official site for comprehensive instructions and compliance tips.
Challenges in Medical Device Disposal
Several challenges complicate the disposal process:
- Identifying Contaminated Devices: Proper identification is essential to ensure that all hazardous devices are safely separated and processed for destruction.
- Cost Management: Disposal requires investment in appropriate equipment, staff training, and logistics, making it one of the more costly operational areas for healthcare providers.
- Adapting to Regulation Changes: Regulations governing destruction and disposal evolve, requiring ongoing education and process improvements to avoid lapses in compliance.
These hurdles require a proactive approach, from adopting the latest best practices to participating in industry training sessions aimed at compliance and safety.
Final Thoughts
Safe, compliant medical device destruction protects public health, ensures the confidentiality of sensitive data, and helps reduce the environmental impact of healthcare operations. By fully understanding and adhering to best practices in device disposal, healthcare facilities can meet their ethical and legal obligations while supporting a cleaner, safer environment for everyone.

